
CEN-ISO/TS 82304-2 was an assignment by the European Commission to the European Committee for Standardization (CEN). The initiative went global in its cooperation with the International Organization for Standardization (ISO). CEN-ISO/TS 82304-2:2021 Health software – Part 2: Health and wellness apps – Quality and reliability was published in July 2021. The core content of this Technical Specification (TS) is a health app quality assessment framework. The results are communicated in a score, a label inspired by the very effective EU Energy label and a health app quality report. The label provides the results of the health app assessment in one glance. The health app quality report aims to provide the level of detail health care professionals need to recommend a health app and gives insurers a basis for decision-making on reimbursement.
There are over 300.000 health apps. Some strengthen healthcare, prevention and self-management. Others have a negative effect on for instance health, safety and data security. It is hard to impossible to distinguish the good ones from the not so good ones in app stores. Work by the European mHealth hub shows in nine European countries one or more health app assessment frameworks have been developed (1). These frameworks aim to filter out the apps that deliver on what they promise. Yet, most have at most a few dozen assessed apps on display in an app library. The detailed requirements, rigour in assessment, qualifications of app assessors and when the app was last assessed are usually not transparent. Apps that did not make the undisclosed thresholds tend to remain unknown. A recent Nature article that compared health app policies in nine countries concluded none of them have yet succeeded in making health app assessment processes efficient (2).
The WHO compared in 2018 20 frameworks for the EU in search of a common framework [Bradway et. Al. 2018, not public]. The frameworks differed quite a lot in topics covered, stakeholders addressed, existing frameworks referenced, and standards aligned with. In its Digital Single Market strategy, the European Commission aims to have digital tools and data contribute to person-centred healthcare and citizen empowerment. Yet, a multitude of frameworks creates an unworkable situation for app manufacturers and proves counterproductive in achieving efficiency, trust, uptake and funding of health apps. The potential of health apps in sustaining healthcare budgets, improving quality and accessibility of healthcare and addressing the shortage in healthcare staff are heavily underutilised. CEN-ISO/TS 82304-2 outperforms all the 20 frameworks the WHO assessed in all criteria used
The assessment framework consists of 81 questions. Sixty-seven of these questions are score-impacting quality requirement questions. To obtain the label for a specific health app, app manufacturers must provide supporting evidence for all the requirements they claim to comply with. This evidence will be assessed by accredited app assessment organisations using the 82304-2 EU certification scheme (‘handbook for app assessors’) that Label2Enable will develop.
Several stakeholders can benefit from CEN-ISO/TS 82304-2:
Health app manufacturers who want to create quality apps
Patients, citizens, and carers who want to use health apps
Health care systems and authorities who want to review, procure or reimburse apps
Health care providers and health care professionals who want to recommend or prescribe apps
App checkers or certification bodies who want to use a trusted, globally recognised health app assessment framework
App stores and app libraries who want to help their customers make informed decisions on health apps
Label2Enable engages with all the above stakeholders. Except for the app stores, they are also represented in our consortium. Would you like to engage too? Subscribe to our newsletter, comment on our personas, follow us on LinkedIn, Twitter or facebook, or email us.
It can be downloaded on the ISO website at a fee.
Annex A of CEN-ISO/TS 82304-2 documents in detail the characteristics of the health app quality label. Translations are currently available in 26 languages. You can find the label translations in our results section. More languages are envisioned. The current translations show the label can be used with different languages, characters and script directions. Label2Enable will provide translations of the label in the official EU languages.
The Label2Enable project tested the label in four corners of Europe with people with low health literacy. With these tests, we learned how to introduce the label adequately.
Please contact us via our pre-order form. We will get in touch with you!
Label2Enable has planned to publish 14 academic publications until the end of 2024, with some already available in the results section.
The available articles include "What Makes a Quality Health App—Developing a Global Research-Based Health App Quality Assessment Framework for CEN-ISO/TS 82304-2: Delphi Study" (2023) by Petra Hoogendoorn (LUMC), "Assessing Health Apps: A Methodology for Health App Quality Evaluation Using the CEN-ISO/TS 82304-2 Framework" by John Smith, and "The Impact of Standardized Health App Assessment on Healthcare Delivery" by Maria Garcia.
The website will be updated as soon as more publications are available.
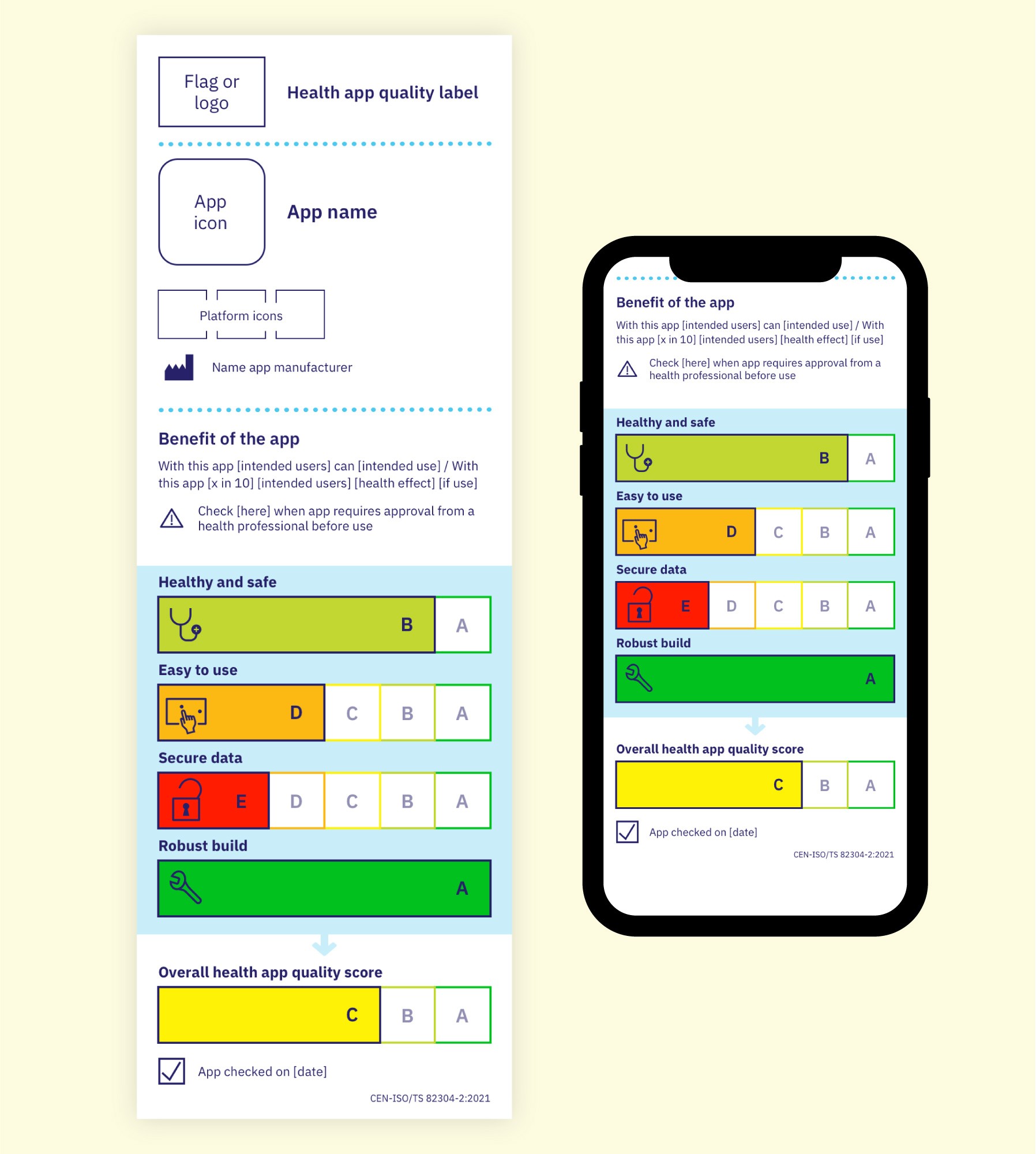
Display of the label
Want to stay up to date?